|
The total mass of all products of a chemical reaction is equal to the total mass of all reactants of that reaction. 
His famous disciple, Democritus Biography & Facts - Britannica of Abdera, named the building blocks of matter atomos, meaning literally indivisible, about 430 bce. Finally, we all assume that we have demonstrated the Law of Conservation of Mass. The atomic philosophy of the early Greeks Leucippus of Miletus (5th century bce) is thought to have originated the atomic philosophy. Dalton and others imagined the atoms that composed all matter as tiny, solid spheres in various stages of motion. Dalton’s atomic theory contains five basic assumptions: All matter consists of tiny particles called atoms. 2.2: The Discovery of Atomic Structure is shared under a CC BY-NC-SA 3.0 license and was authored, remixed, and/or curated by LibreTexts. It was the English chemist, John Dalton, who put the pieces of the puzzle together and developed an atomic theory in 1803. The elements are not transmutable: one element cannot be converted into another. Three important kinds of radiation are particles (helium nuclei), particles (electrons traveling at high speed), and rays (similar to x-rays but higher in energy). For example, metallic iron and gaseous oxygen are both elements and cannot be reduced into simpler substances, but iron rust, or ferrous oxide, is a compound which can be reduced to elemental iron and oxygen. Atomic theory originated as a philosophical concept in ancient India. All other pure substances, which we call compounds, are made up from these elements and can be decomposed into these elements. A Brief History of Atomic Theory The Atom and Atomism. We will assume that we have identified all of these elements, and that there are a very small number of them. We will begin by assuming that all materials are made from elements, materials which cannot be decomposed into simpler substances. However, one of his underlying assumptions was later shown to be incorrect. There are over 18 million known substances in our world. Daltons ideas proved foundational to modern atomic theory. For example, copper and chlorine can form a green, crystalline solid with a mass ratio of 0.558 g chlorine to 1 g copper, as well as a brown crystalline solid with a mass ratio of 1.116 g chlorine to 1 g copper.\) 
He believed all substances were made of small amounts of these four elements of matter. He thought that all materials on Earth were not made of atoms, but of the four elements, Earth, Fire, Water, and Air. The law of multiple proportions states that when two elements react to form more than one compound, a fixed mass of one element will react with masses of the other element in a ratio of small, whole numbers. born on 384 BC died on 322 BC Aristotle did not believe in the atomic theory and he taught so otherwise. For example, there are many compounds other than isooctane that also have a carbon-to-hydrogen mass ratio of 5.33:1.00.ĭalton also used data from Proust, as well as results from his own experiments, to formulate another interesting law. That is, samples that have the same mass ratio are not necessarily the same substance. 3.1: Atomic Theory is shared under a not declared license and was authored, remixed. Each element has its own atomic number, which is equal to the number of protons in its nucleus. Atoms themselves are composed of protons, neutrons, and electrons. 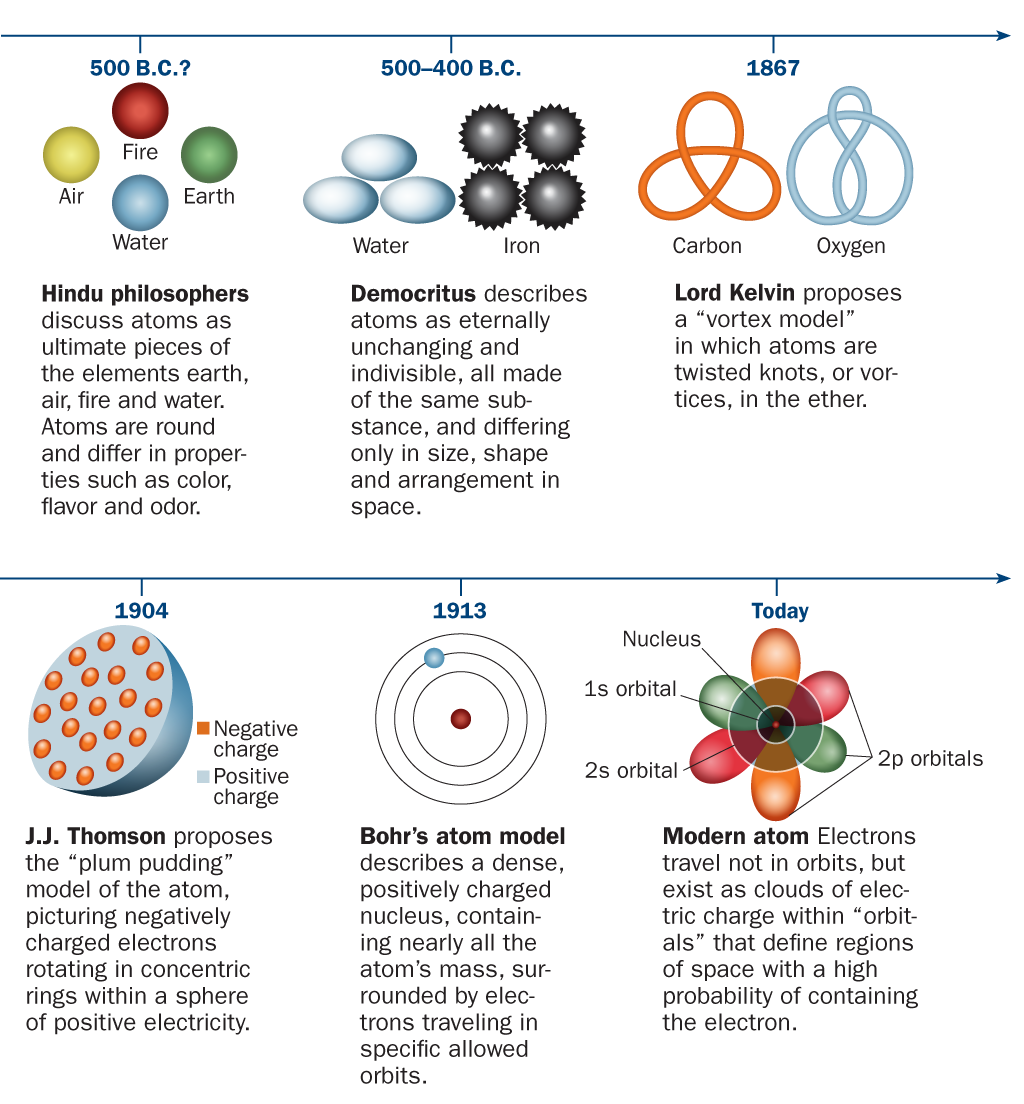
It is worth noting that although all samples of a particular compound have the same mass ratio, the converse is not true in general. Chemistry is based on the modern atomic theory, which states that all matter is composed of atoms.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
